The global demand for GLP-1 and GIP receptor agonists—the blockbuster medications transforming obesity and diabetes management—has created unprecedented challenges for healthcare supply chains. For years, the bottleneck hasn’t just been manufacturing the drug itself, but producing the millions of single-use auto-injector pens required to deliver it.
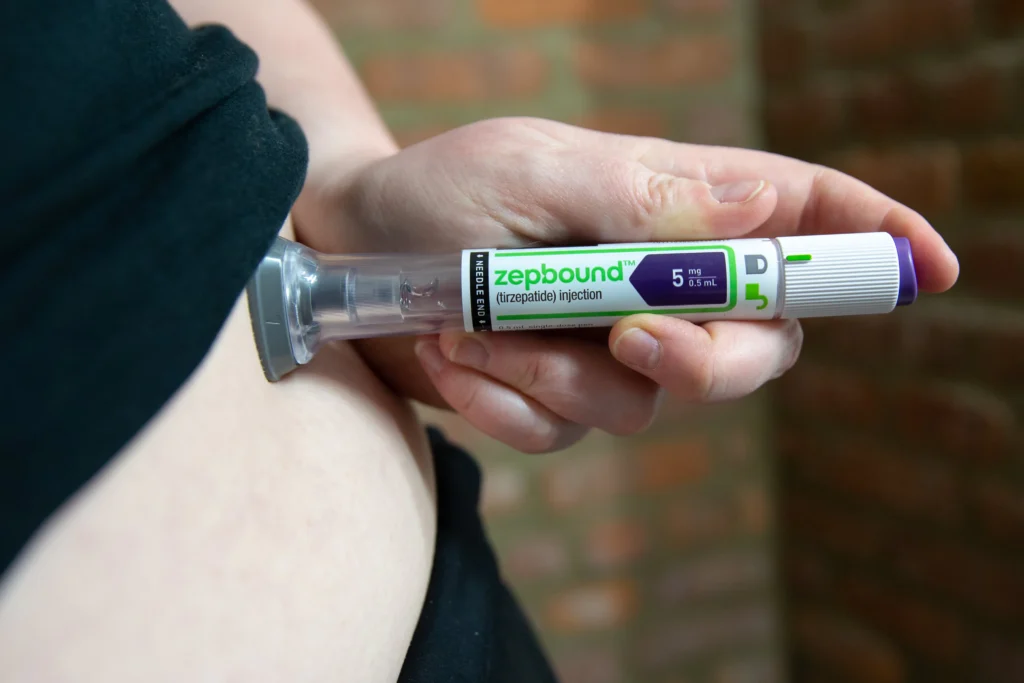
Now, a major device innovation is reshaping the landscape. Eli Lilly has officially launched a multi-dose version of its highly popular weight management drug, Zepbound (tirzepatide), condensing a full month’s supply into a single KwikPen.
Here is why this shift from single-use to multi-dose delivery is a game-changer for patients, healthcare providers, and the global medical supply chain.
The Evolution of the Zepbound KwikPen
Until this latest FDA-approved rollout, patients taking Zepbound either had to use a new, single-dose auto-injector pen every week or manually draw the medication from a vial using a syringe. While effective, the single-use pens required massive amounts of packaging, took up significant cold-storage space, and created a large footprint of medical waste.
The new KwikPen simplifies this process entirely. A single reusable device now holds four weekly doses. Patients simply dial their prescribed dose, attach a fresh needle, and inject. The device is available across all six dosage levels (from 2.5 mg up to 15 mg).
Tackling “Injection Fatigue” and Patient Adherence
Managing a chronic condition like obesity requires long-term medication adherence, but patients often experience “injection fatigue.” Managing four separate pens a month, tracking multiple boxes, and constantly disposing of large auto-injectors can make treatment feel like a burdensome chore.
By streamlining the delivery mechanism into one familiar, multi-dose device, the daily logistics of weight management are simplified. For international patients or those traveling for medical care, carrying a single, discreet pen for a month-long trip is infinitely more manageable than traveling with a bulky cooler full of single-use injectors.

Easing the Global Supply Chain
From a medical equipment and pharmaceutical distribution standpoint, this is a massive leap forward. Consolidating four doses into one device drastically alters the unit economics of healthcare delivery:
- Reduced Packaging: Less secondary packaging means lower manufacturing costs and less environmental waste.
- Streamlined Distribution: Fewer individual pens simplify throughput planning for manufacturers and improve inventory turnover metrics for pharmacies.
- Cold Chain Efficiency: A single month-long pen takes up a fraction of the space in temperature-controlled shipping and storage compared to four individual auto-injectors, making global export and distribution significantly more efficient.
Direct-to-Consumer Pricing Shifts
Perhaps just as disruptive as the device itself is how it is being sold. Bypassing traditional insurance hurdles, Eli Lilly is offering the multi-dose KwikPen directly to self-paying consumers through its LillyDirect platform.
Starting at $299 per month for the initial 2.5 mg dose (and capping at $449 for higher maintenance doses), the pricing aligns with recent pushes to make critical medications more affordable. This direct-to-patient model, combined with a simpler delivery device, removes friction from the purchasing process and puts more control back into the hands of the consumer.
The Takeaway
The launch of the Zepbound multi-dose KwikPen proves that the next phase of the obesity drug boom won’t just be won by the clinical efficacy of the molecule—it will be won by device innovation. By making the medication easier to manufacture, cheaper to distribute, and simpler for the patient to use, the healthcare industry is taking a massive step toward sustainable, long-term weight management solutions.
The Clinical Benefits: Why Zepbound Stands Out
Zepbound is not just another weight-loss fad; it represents a major leap forward in metabolic medicine. Unlike earlier medications that only target one hunger-regulating hormone, Zepbound (tirzepatide) is the first approved “dual-agonist.” It mimics two naturally occurring incretin hormones in the body: GLP-1 and GIP.
This dual-action approach delivers several profound clinical benefits:
- Unprecedented Weight Loss: In clinical trials, patients taking the highest doses of Zepbound lost an average of 20.9% of their body weight (often up to 48–50 pounds) over 72 weeks. This level of weight reduction was previously only seen with bariatric surgery.
- Appetite and Craving Control: By slowing down gastric emptying (how fast food leaves the stomach) and signaling the brain’s satiety centers, Zepbound dramatically reduces the physical feeling of hunger and quiets “food noise” or constant cravings.
- Metabolic Reset: Beyond the scale, the medication improves how the body breaks down sugar and fat. Patients consistently see marked improvements in blood pressure, cholesterol levels, and insulin sensitivity.
- Relief for Sleep Apnea: In a massive breakthrough, the FDA recently approved Zepbound as the very first prescription medication to treat moderate-to-severe Obstructive Sleep Apnea (OSA) in adults with obesity, significantly reducing nighttime breathing disruptions.
Who is Zepbound Actually For?
Despite the hype on social media, Zepbound is not a cosmetic drug for dropping a quick five pounds before a vacation. It is a serious, long-term medical treatment designed for chronic weight management.
According to FDA guidelines, Zepbound is specifically indicated for:
1. Adults with Obesity Individuals who have a Body Mass Index (BMI) of 30 or higher qualify for the medication, regardless of whether they have other underlying health conditions.
2. Adults with Overweight Plus a Comorbidity Individuals with a BMI of 27 or higher (classified as overweight) qualify if they also suffer from at least one weight-related medical condition. These conditions typically include:
- Type 2 diabetes
- Hypertension (high blood pressure)
- Dyslipidemia (high cholesterol)
- Cardiovascular disease
- Obstructive sleep apnea
3. The Commitment to Lifestyle Changes Clinically, Zepbound is prescribed as an adjunct therapy. This means it is intended for individuals who are actively committed to using the medication alongside a reduced-calorie diet and increased physical activity to achieve sustainable, long-term health.